|
If you compare Table 2.1 "Elemental Composition of Earth" and Table 2.2 "Elemental Composition of a Human Body", you will find disparities between the percentage of each element in the human body and on Earth. Table 2.2 "Elemental Composition of a Human Body" lists the relative abundances of elements in the human body. Table 2.1 "Elemental Composition of Earth" lists the relative abundances of elements on Earth as a whole and in Earth’s crust. Hydrogen, the most abundant element in the universe, makes up only 0.14% of Earth’s crust. Oxygen makes up 46.1% of the mass of Earth’s crust (the relatively thin layer of rock forming Earth’s surface), mostly in combination with other elements, while silicon makes up 28.5%. On the planet Earth, however, the situation is rather different. All other elements are present in relatively minuscule amounts, as far as we can detect. In the universe as a whole, the most common element is hydrogen (about 90% of atoms), followed by helium (most of the remaining 10%). Their role is central to chemistry, for they combine to form the millions and millions of known compounds. You may have already figured out that the chemical alphabet consists of the chemical elements. 
However, the chemical alphabet is larger than the one we use for spelling. Just as a language has an alphabet from which words are built, chemistry has an alphabet from which matter is described. (Another important advance was the eradication of polio.) The nationwide reduction of tooth decay has been cited as an important public health advance in history. 
This intentional fluoridation, coupled with the use of fluoride-containing toothpastes and improved oral hygiene, has reduced tooth decay by as much as 60% in children. Currently about 50% of the US population drinks water that has some fluorine added (in the form of sodium fluoride, NaF) to reduce tooth decay. We know now that fluorine combines with hydroxyapatite to make fluorapatite, which is more resistant to acidic decay than hydroxyapatite is. Tooth enamel consists mostly of a mineral called hydroxyapatite, which is composed of calcium, phosphorus, oxygen, and hydrogen. The protective effects of fluorine have a simple chemical explanation. Research continued, and in the 1930s, the US Public Health Service found that low levels of fluorine in water would provide the benefit of resisting decay without discoloring teeth. After years of study, excess fluorine compounds in the drinking water were discovered to be the cause of both these effects. In the early 1900s, a dentist in Colorado Springs, Colorado, noted that many people who lived in the area had brown-stained teeth that, while unsightly, were surprisingly resistant to decay. 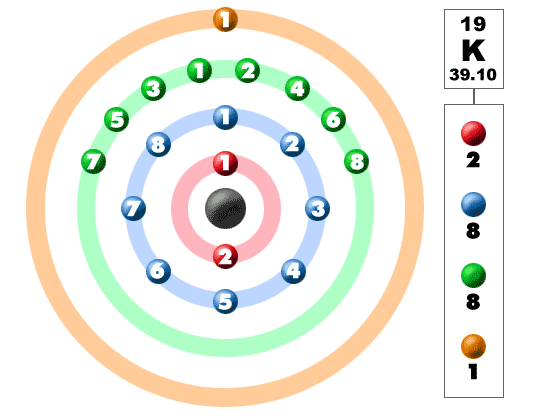
Unprotected by enamel, a tooth will start to decay, thus developing cavities and other dental problems. Acids found in some foods or made by bacteria that feed on food residues on our teeth are capable of dissolving enamel. It has to be hard so that our teeth can serve us for a lifetime of biting and chewing however, tough as it is, tooth enamel is susceptible to chemical attack. (Credit: Christopher Auyeung Source: CK-12 Foundation License: CC BY-NC 3.The hardest material in the human body is tooth enamel. \): Atomic radii of the representative elements measured in picometers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
